Regardless what your opinions are on vaccinations, no matter where you stand on the spectrum from ‘Bill Gates wants to end the world,’ to ‘Coronavirus will kill anyone who isn’t wearing PPE (personal protective equipment), here are some interesting facts you need to know about the leading candidates for the Covid-19 vaccine, “vaccination production” and the different types of vaccinations being made.
Key points:
- The “average” vaccine takes 10-15 years to make
- The fastest vaccine to ever be fully developed took 4 years
- Covid-19 vaccine clinical trials are being fast-tracked
- There is currently no commercial vaccine available for the other coronaviruses (MERS or SARS) to date
- mRNA and DNA vaccines have never been approved for human use
Australia’s long-term plan for dealing with Covid-19
As the Coronavirus pandemic has made its way across the globe, each country has dealt with it in distinct ways by introducing varied restrictions and management plans. Some plans have potentially worked better than others, yet only time will truly tell whether wholesale lockdowns or no lockdowns are effective, and which have had the best cost: benefit outcomes for their citizens and economies.
After some of the recent announcements by Australian politicians this week, it would seem that, directly and indirectly, politicians are threatening that Australians will inevitably have to wait for a vaccine until their lives may return back to any form of normality. NSW Premier Gladys Berejiklian says social distancing measures will remain a fact of life amid the coronavirus pandemic until a vaccine is found even though the number of new cases continues to stabilise.
“This is the new normal,” she said this week, “Not having physical contact, socially distancing is, at the very minimum, a way of life for us. Because we have to be honest with ourselves – until a vaccine is found, there is no simple way to deal with this issue.”
Daniel Andrews, the Victorian Premier, also mentioned in a press briefing on the 07 July 2020 that “until there is a vaccine or a drug or a cure, there is no such thing as “normal”. He reiterated that again on the 24th August by saying that the State of Emergency will be in place for 18 months and lockdown restrictions will be extended beyond September. This is despite the state recording its lowest rise in COVID-19 infections in seven weeks. Adding, “If there is no vaccine within 12 months, the state’s restrictions may have to be extended again.”
Australia’s prime minister has said that he wants to make any potential Covid-19 vaccine as mandatory as possible. Scott Morrison said the aim was to get 95% of the population to have the jab, “I would expect it to be as mandatory as you can possibly make,” he said in a radio interview.
Nancy Baxter, the head of Melbourne University’s School of Population and Global Health, said Victorians would need to get used to long-term lockdown.“I’m not sure if we will ever get beyond stage 2, I think we will always be in stage 2.2, a stage between 2 and 3 until we get a vaccine,” she said.
It is evidently clear that the approach of the Australian government and similar countries such as New Zealand is heavily dependent upon the development of a vaccine for life to return to any form of normal.
So let’s take a quick look at the process for vaccine production and why some people are alarmed by this…
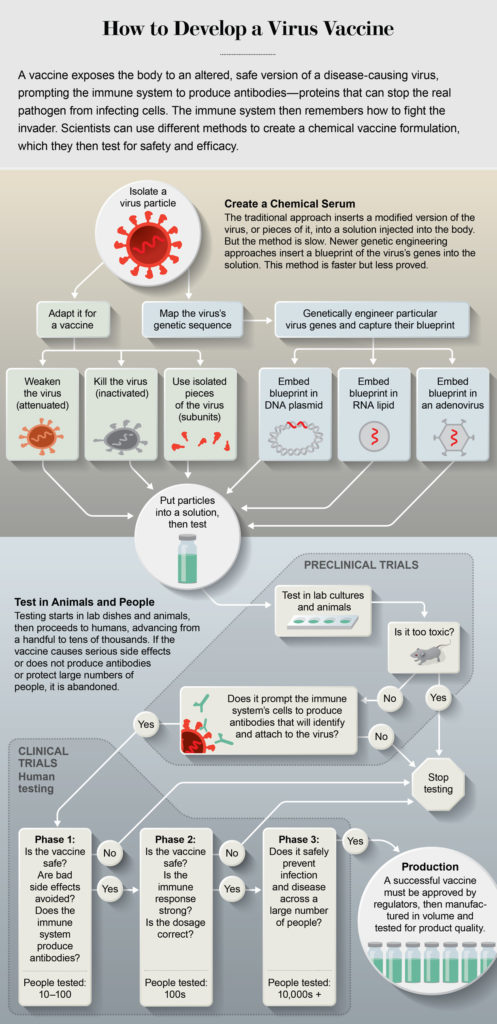
 Here is a diagram summary explaining part of this development cycle, for a more in-depth explanation of this process please visit here.
Here is a diagram summary explaining part of this development cycle, for a more in-depth explanation of this process please visit here.
Vaccine production
How long does the average vaccine take to make?
According to historyofvaccines.org “Vaccine development is a long, complex process, often lasting 10-15 years and involving a combination of public and private involvement.”
How fast was the fastest vaccine ever made?
The mumps vaccine considered one of the fastest ever approved, took four years to go from collecting viral samples to licensing a drug in 1967.
The next fastest vaccine development was the Ebola vaccine ‘Erbivo’ released earlier this year by MSD after Ebola vaccine development began in 2014.
The main reason vaccine production takes so long is because they must undergo a series of testing stages until they become approved for clinical use.
The general stages of the development cycle of a vaccine are:
- Exploratory stage
- Pre-clinical stage
- Clinical development (Phase 1, 2 and 3)
- Regulatory review and approval
- Manufacturing
- Quality control
Covid-19 Fast Tracking
Due to many countries deciding that a return to normal life depends upon a COVID-19 vaccine, the clinical trial process for such a vaccine may be cut short.
Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, said in April that a promising COVID-19 vaccine that prompts a strong immune response in Phase 2 trials could potentially be approved for emergency use without a full Phase 3 trial, Live Science previously reported. Phase 3 trials for vaccines typically last several years, according to the CDC, but given the “pressing” need, a coronavirus vaccine could potentially be granted emergency approval in less time, Fauci said.
Other potential issues:
The genetic stability of the virus obviously matters as well. Some viruses, such as influenza, mutate so rapidly that vaccine developers must release new formulations each year. Another example would be HIV which has a rapid evolution and this a major reason we still have no vaccine for the disease.
So far, the Sars-CoV-2 coronavirus seems fairly stable, but it is acquiring mutations, as all viruses do. Some genetic changes have been spotted in the virus’s protein “spikes” which are the basis of most vaccines. If the spike protein mutates too much, the antibodies produced by a vaccine will effectively be out of date and might not bind the virus effectively enough to prevent infection.
Martin Hibberd, professor of emerging infectious diseases at the London School of Hygiene and Tropical Medicine, who helped identify some of the virus’s mutations, called them “an early warning”.
What we can learn from the other Coronaviruses.
Coronaviruses are a group of viruses that belong to the family Coronaviridae. In humans, coronavirus infections have been shown to be potentially lethal. This is the case with severe acute respiratory syndrome (SARS) originating in southern China 2002–2003, and the Middle East respiratory syndrome (MERS) coronaviruses originating in Saudi Arabia in September 2012. (Well at least we all know what these abbreviations finally mean now?)
There is currently no commercial vaccine available for MERS or SARS to date.
Padron-Regalado E. Vaccines for SARS-CoV-2: Lessons from Other Coronavirus Strains [published online ahead of print, 2020 Apr 23]. Infect Dis Ther. 2020;9(2):1-20. doi:10.1007/s40121-020-00300-x
What have we learned from trying to develop vaccines for these viruses?
During the search for a Sars vaccine in 2004, scientists found that one candidate caused hepatitis in ferrets.
Another serious concern that arose was “antibody-induced enhancement” where the antibodies produced by a vaccine actually make future infections worse. The effect caused serious lung damage in animals given experimental vaccines for both Sars and Mers.
 Who is Leading the Covid-19 Vaccine Race?
Who is Leading the Covid-19 Vaccine Race?
The Regulatory Affairs Professionals Society currently has a COVID-19 vaccine tracker available on their website that you can visit here and look at each of the vaccine company contenders. This is a great resource for when you hear a vaccine brand name on the news. This website helps you ascertain who’s funding the development and at what phase of development, the vaccine is currently at.
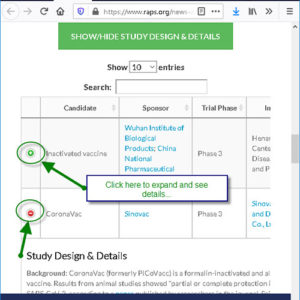 The Australian government has confirmed an agreement was reached with British pharmaceuticals giant AstraZeneca to secure 25 million doses of the vaccine being developed by Oxford University if it proves successful in human trials. Interestingly, AstraZeneca as a pharmaceutical company has a terrible track record with an incredible number of legal disputes regarding product safety.
The Australian government has confirmed an agreement was reached with British pharmaceuticals giant AstraZeneca to secure 25 million doses of the vaccine being developed by Oxford University if it proves successful in human trials. Interestingly, AstraZeneca as a pharmaceutical company has a terrible track record with an incredible number of legal disputes regarding product safety.
While the US government has also chosen three of these vaccine candidates to fund for Phase 3 trials under Operation Warp Speed: Moderna’s mRNA-1273, The University of Oxford and AstraZeneca’s AZD1222, and Pfizer and BioNTech’s BNT162. Two of these vaccines that the US government has chosen to fund, are Moderna’s mRNA-1273 and BioNTech’s BNT162 are mRNA vaccines.
How Do These Newly Proposed Vaccines Differ from Vaccines Currently Available?
The most traditional methods of vaccine development that we currently have; introduce the body to either an inactivated or weakened form of a virus or to one of its viral proteins.
The immune system responds by producing antibodies that recognise particular proteins of the virus. The intention then is that those antibodies could fight future infections, as long as the virus doesn’t evolve.
While the intention of vaccines at its core has always been noble, many people have questions around the safety of the active and added ingredients in vaccines and the lack of safety studies on vaccines to date. Alarm bells are heightened than when discussions on new types of vaccines are being pushed particularly at an accelerated rate.
So what is an mRNA vaccine then?
mRNA vaccines take a novel approach that has never been licensed for human use.
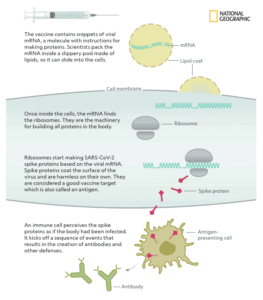 Potential Adverse Effects and Risks:
Potential Adverse Effects and Risks:
- The mRNA strand in the vaccine may elicit an unintended immune reaction. To minimise this, the mRNA vaccine sequences are designed to mimic those produced by mammalian cells (for example monkey cells).
- A possible concern could be that some mRNA-based vaccine platforms induce potent type I interferon responses, which have been associated not only with inflammation but also potentially with autoimmunity. Thus, identification of individuals at an increased risk of autoimmune reactions before mRNA vaccination may allow reasonable precautions to be taken.
So what is a DNA vaccine?
The ZyCoV-D vaccine sits on this top ten most likely Covid-19 vaccine candidate list, this is a DNA vaccine.
DNA vaccines work very similar mRNA vaccines, however instead of containing mRNA strands, DNA vaccines contain bacterial plasmid DNA genetically modified to contain the nucleotides, encoding an antigenic portion of the virus such as the viral core region or envelope region. The DNA is taken up into the host cell’s nucleolus as opposed to the mRNA vaccine which only travels to the cell cytoplasm.
Potential Adverse Effects and Risks:
- Initial attempts to create DNA vaccines have not worked – they have not had a big enough impact on the immune system.
- No DNA vaccine has been licensed for use in humans yet – although some DNA vaccines are now in clinical trials, none are licensed for use – so they are an unproven method.
I spend much of my time reading journal articles and I am always enthusiastic to see scientific breakthroughs. With these new forms of vaccines, I am, however, particularly dubious about them being fast-tracked, especially since safety around our current vaccines is a large concern for many. Rushing new vaccines at the height of a pandemic seems to only heighten alarm bells.
The Russian Vaccine
In his most recent announcement, Putin has said that the Russian regulator had approved a COVID-19 vaccine developed by the Gamaleya Research Institute of Epidemiology and Microbiology in Moscow, even though phase III trials of the vaccine had yet to be completed.
The Gamaleya vaccine (a non-replicating viral vaccine) has been given to 76 volunteers as part of two early-stage trials listed on ClinicalTrials.gov, but no results from those trials or other preclinical studies have been published, and little else is known about the experimental vaccine.
Experts Worry Vaccine Uptake May Be Low
Are you still considering getting the Covid-19 vaccine when it comes out?
If you aren’t, if you are hesitating, you definitely wouldn’t be alone. An Australian poll involving 77,200 voters showed that 67% of Australians would not choose to take the COVID-19 vaccine when it becomes available. Read more about this by clicking on this link (accessed and updated as of 12:00pm 20/08/20).
It seems many Australians would like to hold off until they feel more confident that the safety of this vaccine is not being compromised in its manufacturing process. At any rate, they would like to be given the choice as to whether they agree to take the vaccine versus the vaccine being mandated or experiencing repercussions for delaying their uptake.
How Do We Move Forward from Here?
I’m sure we have all been keeping our eye on other countries such as America, New Zealand, and Sweden to see how they are handling the pandemic. We all have varying opinions on which of these are working the most effectively, definitely a conversation for another day. However, I would like to touch on some Australian statistics that are not getting as much attention as they potentially should.
You may remember that back when China and Europe were sent into a frenzy initially with this virus and as initial cases rose in Australia, we were all willing to do “the right thing” and stay at home so as to not potentially increase the spread of the virus to the vulnerable and elderly in our communities. So as to not overload our hospital systems.
The overarching goal of the restrictions and public health measures was to ensure that our countries healthcare system did not collapse as they had in other countries that were hit hard by the virus. Making sure we had enough ICU beds for example for those who needed them was obviously very important, so one question to ponder is – why haven’t we been talking about the current state and capacity of our healthcare system, instead of solely focusing on how many people have tested positive for COVID-19?
Interestingly do you know many ICU beds do we have in Australia?
And how many Covid-19 patients do we have in ICU’s?
Only 44 (as of 19/08/20)
Obviously other factors come into play when assessing the burden on health care systems, but surely this ratio should be part of the conversation on the incidence and the impact of COVID-19, rather than solely making alarmist comments from Premier Andrews for example that, “It’s upsetting that we have had another 500 cases today”. Using case numbers versus death rate has been in question throughout this pandemic.
According to the ‘IMPACT OF COVID-19: theoretical modeling of how the health system can respond’ document that was published by the Australian government at the beginning of the pandemic, “With a 33% reduction in transmission due to social distancing, the proportion of people infected is 12% and only 5% require some medical care. Everyone who needs an ICU bed over the course of the pandemic could access one. As of 20/08/20, there are 7,434 active Covid-19 cases in Australia from a total national population of 24.99 million people. This means that we currently have a 0.0297% proportion of people infected (well below 12%). If we use this model, we are currently well away from having a healthcare system collapse on our hands.
No one leader or political party has all the solutions or answers, and healthcare workers are doing an amazing job at keeping our country safe as they try to follow top-down directives. Politicians are also trying to navigate these unknown set of circumstances and the “COVID-19 terrain.” AUSTRALIAN politicians, however, have fallen under heavy criticism with their seemingly inadequate hotel quarantine protocols and heavy-handed lockdowns that have shattered the livelihoods on citizens and our nation’s economy.
It also seems potential futile to force Australia to wait for a vaccine to arrive before our lives can return to normal and restrictions be lifted.
A vaccine is not our only option particularly when we consider the studies that demonstrate that Ivermectin and hydroxychloroquine may be cheaper and safer, viable options for preventing and treating this virus. Surely our government could give both long-term medications some consideration rather than blatantly ignoring them.
One thing that we can be sure of – is that a strong immune system offers us the best chance we have to fight this virus and topic and theme that has not been emphasized anywhere near enough by our government or media.
We hope this blog has helped answer some of your questions about how these new vaccines may work.
Yours in Health
Jennifer Barham-Floreani,
Bach. Chiropractic, Bach. App Clinical Science
Registered internationally, no longer practicing as a chiropractor in Australia.
Wilem Floreani,
Research Assistant & Student
Barcelona College of Chiropractic



3 thoughts on “Waiting For A Cure…”
Great work guys in putting this together ! Champions !!
Thanks for a concise article and explanation about the development process. Excellent food for thought and deliberation.
Good on you! We need more of your kind( who CARE !) God. Bless you and keep up the fight for truth, politician s need to be held accountable, also police, health professionals , and ALL who are supposed to have our ( the people). Best interests @ heart. I am so very impressed with you and shall forward this on.